Tech Story
ABSTRACT
Joining dissimilar materials, especially cladding highquality material over a lowquality substrate surface for multifunctionality applications, is one of the potential research interests. Aluminium and stainless steel are the two predominantly used engineering materials in this realm. Nevertheless, cladding is yet to be explored while a substrate is being shaped into a product. This study aims to clad the austenitic stainless steel to a hypo-eutectic aluminium alloy through die-casting via reverse-cladding procedure by keeping the prepared stainlesssteel sheet inserted in the die cavity and pouring the aluminium-silicon molten metal over it. Microscopic studies and the electron probe microanalysis showed the formation of distinct intermetallic layers of variable thickness across the cladding region. The outcomes disclosed that the reverse cladding could be achieved successfully through the casting route.
INTRODUCTION
Joining similar or dissimilar materials for wear, corrosion, and tribological applications is of high research interest. It is due to its ability to increase the strength-to-weight ratio and overall cost of the component with good functionality, which may not be economically achieved with a single material. Aluminium has favourable ductility, corrosion resistance, and thermal and electrical conductivity at low temperatures. In contrast, austenitic stainless steel possesses excellent corrosion resistance and mechanical properties at elevated as well as cryogenic temperatures. Dissimilar joints between these two can be applied in cryogenic pipe couplings and fuel storage vessels due to their properties
Researchers have produced dissimilar aluminium and stainless-steel joints using arc welding, arc welding-brazing, double-sided arc welding-brazing, friction welding, friction-stir welding, impact welding with/without an interlayer, laser welding, and wire-arc direct energy deposition process. However, all these processes have certain limitations. Arc welding leads to the formation of thick intermetallic layers, and most of these processes are shape and size-restricted processes. The successful joinability of aluminium and stainless steel through the explosive cladding technique could be achieved if the aluminium plate is used as a flyer
One advantage of dissimilar joining of the aluminum and austenitic stainless steel is that joining is diffusion controllable. It was experimentally found that the diffusion of iron, chromium, and nickel from austenitic stainless steel in molten aluminum at 700 to 850OCis non-selective and diffusion-controlled [3]. Some researchers found ways to reduce intermetallic layer thickness during the dissimilar joining of these two materials during arc welding-brazing processes. Experimental investigations have shown that low silicon content reduces the intermetallic layer thickness by restricting the iron dissolution rate [4]; copper addition forms a thin crack-resistant intermetallic layer due to the formation of aluminium-copper strong bonds through the usage of aluminium- silicon and aluminium-copperfiller materials, respectively, while joining aluminium alloys to stainless-steel through tungsten inert gas welding-brazing [5]. A double-side metal inert gas-tungsten inert gas arc welding-brazing process formed a thinner intermetallic layer than the conventional welding-brazing for similar heat input conditions [6]. However, all these techniques are utilised on the component already shaped using any primary shaping processes. Furthermore, joining dissimilar materials, especially cladding a suitable material on a component while it is being fabricated, needs attention. Previous studies indicate that the joining of these two materials is possible through welding, so it might be possible to join them through its sister process, i.e., casting. So, this study aims to address the reverse clad of austenitic stainless steel to the hypo-eutectic aluminium-silicon alloy while it is being cast. The reverse cladding procedure involves the deposition of the substrate material on the surface-prepared sheet insert of source material, which is exactly the reverse of the conventional cladding procedure.
MATERIALS AND METHODOLOGY
The presence of low silicon results in thinner intermetallic layer formation at the interface when the aluminum alloy is joined to stainless steel through welding. Nevertheless, low silicon-containing hypo-eutectic alloys contain a large freezing range, which may result in shrinkage porosity at the interface while joining those two materials through casting via the reverse cladding route. Higher silicon content results in a thicker intermetallic layer, which deteriorates the mechanical properties. Among the primarily castable aluminium-silicon alloys, LM25 contains silicon around 7.5 per cent by weight. The medium silicon content of LM25 alloy, i.e., slightly more than half the concentration of eutectic silicon composition, led to choose it as cast aluminium-silicon alloy for this study along with austenitic stainless steel of 304 grade (Fe-18Cr-8Ni-2Mn) as the sheet insert.
A computerised bottom pouring type stir casting furnace with the die-casting set-up was used to prepare the molten metal. A die with a cavity containing two identical cylinders of 30mm diameter and 300mm length lying on a parallelepiped base of a square cross-section of 900 mm2 and 80mm length. A 2mm thick stainless-steel sheet insert with a length less than the base cavity length by 3mm surface was prepared by emery paper polishing up to 1000 grit size and cleaning with ethanol. The molten metal temperature was set as 925OC. The prepared molten metal with 925±25OC temperature was poured into the die cavity containing sheet insert at its base without preheating. The solidified casting with integrated sheet insert was taken out of the die after complete solidification, followed by cooling up to room temperature.
Samples from the casting with the integrated stainless-steel sheet insert were extracted for characterisation using a precision cutter. Through the standard metallographic procedures, the samples were prepared for metallographic tests. Micrographs of the reaction zone were captured using Leica’s optical microscope and JEOL’s scanning electron microscope for the micrographs. JEOL’s electron probe microanalyser carried out elemental mapping and spectrum analysis.

Fig 1: Optical microscopic micrographs of the reaction zone taken at 200x magnification: a) near edge region and b) near central region
RESULTS AND DISCUSSION
Solidification of the molten metal depends on the heat transfer and the composition. For the parallelopiped portions, solidification starts at the corners, and then the solidification front moves along the edges and finally moves towards the center region. Formation of the reaction and then the corner region. Micrographs of the reaction zone at the edge and central regions were taken using optical and scanning electron microscopes to verify the reaction zone variation. Optical micrographs are shown in Fig 1, and scanning electron micrographs of both regions are in Fig 2.
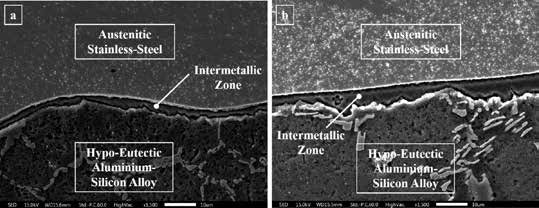
Fig 2: Scanning electron microscopic micrographs of the reaction zone taken at 1500x magnification: (a) near edge region and (b) near central region
The micrographs show that the reaction zone became thicker from the edge region to the central, which supports the previously mentioned statements. However, to verify the non-selective nature of diffusion of elements from austenitic stainless on reaction with molten aluminium alloy, spectroscopic analysis was done across the reaction zone in the form of line scans and represented in Fig 3. The results indicated that the profiles of the iron, chromium, and nickel at the reaction zone replicated each other, confirming the non-selective diffusion nature. The diffusion nature of these three elements supports the previous study.
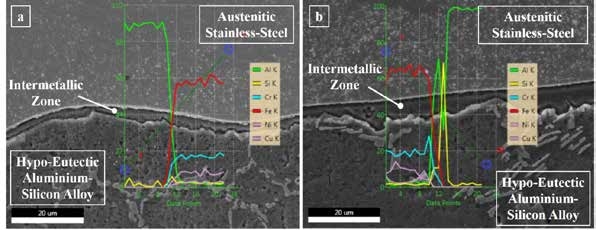
Fig 3: Energy dispersive spectroscopic elemental line mappings: (a) near edge region and (b) near central region
During dissimilar joining, the reaction zone forms due to the inter-diffusion of elements from either side of the materials. From the iron-aluminium phase diagram and the Hume-Rothery rules, it can be observed that body-centered cubic iron can dissolve aluminium in it and form a solid solution. Whereas face-centered cubic aluminium stays approximately as pure aluminium without accepting iron. Due to the limited solubility of aluminium in iron and approximately no solubility of iron in aluminium, a reaction zone consisting of intermetallic layer(s) on inter-diffusion should be formed. The concentration of each element in any intermetallic layer is generally fixed; nevertheless, it may slightly differ from the stoichiometric concentration. To validate the formed reaction zone consisting of intermetallic layer(s), quantitative area mappings were taken using an electron probe micro-analyser, represented in Fig 4. From the color area mapping of aluminium and iron, it can be observed that the formed reaction zone contains different layers, with each containing approximately a fixed concentration of elements; hence, it was confirmed that the formed layers are intermetallic layers.
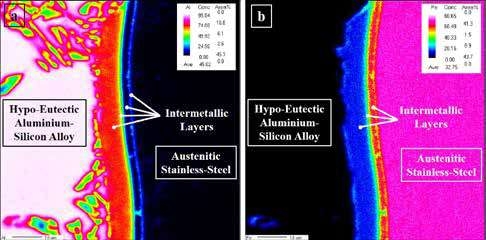
Fig 4: Wavelength dispersive spectroscopic elemental area mappings:(a) aluminium mapping and (b) iron mapping
CONCLUSIONS
With this work, it is experimentally found that reverse-cladding of suitable material to a substrate while it is being cast is feasible if they can form reaction zones on interaction at solid-liquid/solid states, respectively. The reaction zone thickness on the solidification pattern and composition of the molten metal. Rever-cladding of austenitic stainless steel to hypo-eutectic aluminium-silicon alloy resulted in the formation of a reaction zone containing intermetallic layers with its thickness increasing from the edge region to the central region. The reverse cladding results in the same reaction zone formation as the conventional cladding methodologies; however, it eliminates the deposition stage of cladding material on the fabricated component.
References
[1] G. H. S. F. L. Carvalho, I. Galvão, R. Mendes, R. M. Leal, and A. Loureiro, Explosive welding of aluminium to stainless steel using carbon steel and niobium interlayers, J. Mater. Process. Technol., vol. 283, no. October 2019, p. 116707, 2020, doi: 10.1016/j. jmatprotec.2020.116707.
[2] G. H. S. F. L. Carvalho, I. Galvão, R. Mendes, R. M. Leal, and A. Loureiro, Explosive welding of aluminium to stainless steel, J. Mater. Process. Technol., vol. 262, no. March, pp. 340–349, 2018, doi: 10.1016/j. jmatprotec.2018.06.042.
[3] V. I. Dybkov, Interaction of 18Cr-10Ni stainless steel with liquid aluminium, J. Mater. Sci., vol. 25, pp. 3615–3633, 1990.
[4] J. L. Song, S. B. Lin, C. L. Yang, and C. L. Fan, Effects of Si additions on intermetallic compound layer of aluminum-steel TIG welding-brazing joint, J. Alloys Compd., vol. 488, no. 1, pp. 217–222, 2009, doi: 10.1016/j.jallcom.2009.08.084.
[5] J. L. Song, S. B. Lin, C. L. Yang, C. L. Fan, and G. C. Ma, Analysis of intermetallic layer in dissimilar TIG welding-brazing butt joint of aluminium alloy to stainless steel, Sci. Technol. Weld. Join., vol. 15, no. 3, pp. 213–218, 2010, doi: 10.1179/136217110X1266 5048207610.
[6] Y. Zhang et al., Study on MIG-TIG double-sided arc welding-brazing of aluminum and stainless steel, Mater. Lett., vol. 172, pp. 146–148, 2016, doi: 10.1016/j. matlet.2016.02.146.
[7] T. Tirumala, A. Kumar, C.S. Perugu, Reverse cladding of 304 stainless steel to LM25 aluminium alloy through die-casting, Mater. Lett. 349 (2023) 134896.
[8] Ajay Kumar and Tumula Tirumala, A Process for Surface Coating through Casting with Sheet Inserts, Indian Patent, 528628.















